Reusing Titanium Healing Abutments
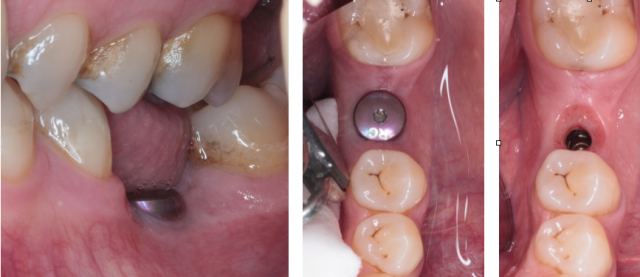
If your practice does anything at all with osseointegrated dental implants, you will be familiar with the role of the healing abutment. Stock titanium healing abutments serve to protect the implant’s prosthetic connection and support the peri-implant soft tissue during healing.
In the United States, it’s common practice to reuse healing abutments, yet little is known about reusing these components. Negative impact on mechanical properties during the cleaning and sterilization process has yet to be studied.
A new study on contamination
Recently, Wadhwani et al. (2015 ahead of print) published a study with the goal of evaluating residual contamination on commercially available titanium healing abutments.
Materials and methods
In this study, researchers collected 100 commercially available healing abutments from eight different dental offices. Seven different implant manufacturers were represented. The abutments were retrieved, cleaned, and sterilized before being collected by the researchers. The manner in which the abutments were cleaned and sterilized varied but followed individual office protocols. Some abutments had been wiped with a disinfectant cloth while others had been cleaned by ultrasonic bath, in water or alcohol, but all the tested abutments were ultimately steam-autoclaved.
The samples were evaluated visually for debris, damage or other contamination, then processed with a stain to detect any residual protein or peptide remnants. Three new, unused titanium healing abutments served as the control group.
Results
Two-thirds (68/100) of the sample healing abutments initially appeared to be free from any residual contamination, but with staining, nearly all (99/100) of the samples were found to have some degree of residual contamination. In contrast, the control group had no staining at all.

What about the one sample healing abutment that didn’t test positive for residual contamination? The authors speculate that this healing abutment was opened but never put into service. In other words, that abutment was probably removed from the packaging but never connected to an implant clinically.
Conclusion
Due to essentially universal contamination of the used, cleaned, and sterilized titanium healing abutments, the authors conclude that the practice of reusing healing abutments must be reconsidered.
What does this mean for clinicians?
Although this is just one article, the implications are critically relevant for all clinicians who use osseointegrated dental implants in their practice. Healing abutments are one of the most commonly used implant components, and are used from the time the implant is placed (in a single-stage approach) to insertion of the definitive abutment. Contaminated healing abutments have the potential to negatively affect peri-implant soft- and hard-tissue healing and maturation.

In addition to potential for biologic problems, it is possible that debris could be transferred from the contaminated healing abutment to the internal surfaces of the implant fixture. Debris inside the fixture could alter the implant/abutment connection, negatively affecting the stability of the connection.
Finally, the risk for cross-contamination must be considered. Contaminated remnants on healing abutments could be a source of disease transmission.
One aspect of this report that stands out is the heterogeneity of cleaning and sterilization protocols, even in the eight dental practices from which the samples were collected. Variation in the approaches to cleaning and sterilizing the components indicates that there isn’t one universally accepted protocol for processing used implant components.
Sodium hypochlorite solutions have been shown to be effective for, if not removing proteins, at least denaturing residual proteins, yet none of the offices reported using this type of solution. In addition, the possible effects of a bleach solution on the mechanical properties of the titanium components remains unclear.
High-intensity LED light set to specific wavelengths is showing promise as a technique for cleaning and sterilizing both air and liquids. This technique holds promise for use with implant components, but a definitive protocol has yet to be determined.
In my opinion, after assessing our current knowledge base, the economic benefit of reusing titanium healing abutments is outweighed by the dangers of residual contamination. This isn’t new, either; Straumann has always recommended using new, original manufacturer components. Several implant manufacturers have programs in place to collect used healing abutments.
Questions? Now would be a good time to reach out to your local implant rep and find out what they recommend. And, as always, it’s a great time to head over to Spear Talk and see how your colleagues are adapting to this new information.
References
- Wadhwani C, Schonnenbaum TR, Audia F, Chung K-H. In-vitro study of the contamination remaining on used healing abutments after cleaning and sterilizing in dental practice. Clinical Implant Dentistry and Related Research. 2015.
- Sennerby L, Lekholm U. The soft tissue response to titanium abutments retrieved from humans and reimplanted in rats. A light microscopic pilot study. Clin Oral Implants Res. 1993; 4(1):23–27.
- Aldosari AAM. Does steam autoclaving affect the accuracy of implant impression systems? Journal of Biomaterials and Tissue Engineering. 2014; (7):550–554.
- Canullo L, Micarelli C, Lembo-Fazio L, Iannello G, Clementini M. Microscopical and microbiologic characterization of customized titanium abutments after different cleaning procedures. Clin Oral Impl Res. 2012; 25(3):328–336.
- Gehrke P, Tabellion A, Fischer C. Microscopical and chemical surface characterization of CAD/CAM zirconia abutments after different cleaning procedures. A qualitative analysis. J Adv Prosthodont. 2015; 7(2):151.
SPEAR Membership
Learn Faster. Treat Better.
Whether you have 10 minutes or time to dive deeper, Spear Membership gives you the learning, tools, and support to keep improving.
Choose the level of support and structure that fits your goals.

By: Darin Dichter
Date: April 4, 2016
Featured Digest articles
Insights and advice from Spear Faculty and industry experts